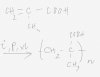Bài 1
a)
[tex]2HCOOH + 2Na \rightarrow 2HCOONa + H_2 [/tex]
[tex]2C_2H_5COOH + 2Na \rightarrow 2C_2H_5COONa + H_2 [/tex]
[tex]2C_2H_5OH + 2Na \rightarrow 2C_2H_5ONa + H_2 [/tex]
[tex]2C_3H_5(OH)_3 + 6Na \rightarrow 2C_3H_5(ONa)_3 + 3H_2 [/tex]
[tex]2CH_2=CH-CH_2OH + 2Na \rightarrow 2CH_2=CH-CH_2ONa +H_2 [/tex]
[tex]2CH \equiv C-COOH + 2Na \rightarrow 2CH \equiv C-COONa + H_2[/tex]
b)
[tex]HCOOH + NaOH \rightarrow HCOONa + H_2O [/tex]
[tex]C_2H_5COOH + NaOH \rightarrow C_2H_5COONa + H_2O [/tex]
[tex]CH_3COOC_2H_5 + NaOH \rightarrow CH_3COONa + C_2H_5OH [/tex]
[tex]HCOOCH_3 + NaOH \rightarrow HCOONa + CH_3OH [/tex]
[tex]CH\equiv C-COOH + NaOH \rightarrow CH\equiv C-COONa + H_2O[/tex]
c)
[tex]CH_2 = CH-CH_2OH + Br_2 \rightarrow CH_2Br-CHBr-CH_2OH [/tex]
[tex]CH \equiv C-COOH + Br_2 \rightarrow CHBr_2 -CBr_2-COOH[/tex] (*)
d)
[tex]2HCOOH + Mg \rightarrow (HCOO)_2Mg + H_2 [/tex]
[tex]2C_2H_5COOH + Mg \rightarrow (C_2H_5COO)_2Mg + H_2 [/tex]
[tex]2CH \equiv C-COOH + Mg \rightarrow (CH\equiv C-COO)_2Mg + H_2[/tex]
e)
[tex]CH_2 = CH-CH_2OH + H_2 \overset{Ni,t^{o}}{\rightarrow} CH_3-CH_2-CH_2OH[/tex]
[tex]CH \equiv C-COOH + H_2 \overset{t^{o},Ni}{\rightarrow} CH_3-CH_2-COOH[/tex]
f)
[tex]HCOOH + NaHCO_3 \rightarrow HCOONa + CO_2 + H_2O [/tex]
[tex]C_2H_5COOH + NaHCO_3 \rightarrow C_2H_5COONa + CO_2 + H_2O[/tex]
[tex] CH\equiv C-COOH + NaHCO_3 \rightarrow CH\equiv C-COONa +CO_2 + H_2O[/tex]
g)
[tex]2CH \equiv C-COOH + Ag_2O \overset{NH_3}{\rightarrow} 2CAg\equiv C-COOH + H_2O[/tex]
Bài 2
[tex]CH_2 = C(CH_3)-COOH + Br_2 \rightarrow CH_2Br-CBr(CH_3)-COOH [/tex]
[tex]CH_2= C(CH_3)-COOH + H_2 \overset{Ni, t^{o}}{\rightarrow} CH_3-CH(CH_3)-COOH [/tex]
[tex]2CH_2=C(CH_3)-COOH + 2Na \rightarrow 2CH_2=C(CH_3)-COONa + H_2 [/tex]
[tex]CH_2 =C(CH_3)-COOH + Cl_2 \overset{xt, t^{o}}{\rightarrow} CH_2Cl-CCl(CH_3)-COOH [/tex] (*)
[tex]2CH_2=C(CH_3)-COOH + Ca(OH)_2 \rightarrow (CH_2=C(CH_3)-COOH)_2Ca + 2H_2O [/tex]
[tex]CH_2=C(CH_3)-COOH + CH_3CH_2OH \overset{H2SO4 dac, t^{o}}{\rightarrow}CH_2=C(CH_3)-COOCH_3CH_2 + H_2O[/tex]
[tex]2CH_2=C(CH_3)-COOH + CaCO_3 \rightarrow (CH_2=C(CH_3)-COO)_2Ca + CO_2 + H_2O [/tex]
[tex]2CH_2 =C(CH_3)-COOH + Mg \rightarrow (CH_2=C(CH_3)-COO)_2Mg + H_2[/tex]
Mấy cái (*) là mình không chắc lắm, bạn coi lại cân bằng giúp mình nhé
Sai thì nói mình xem lại
 [TẶNG BẠN] TRỌN BỘ Bí kíp học tốt 08 môn
[TẶNG BẠN] TRỌN BỘ Bí kíp học tốt 08 môn
 Chắc suất Đại học top - Giữ chỗ ngay!!
ĐĂNG BÀI NGAY để cùng trao đổi với các thành viên siêu nhiệt tình & dễ thương trên diễn đàn.
Chắc suất Đại học top - Giữ chỗ ngay!!
ĐĂNG BÀI NGAY để cùng trao đổi với các thành viên siêu nhiệt tình & dễ thương trên diễn đàn.